Scientists can send a man to the moon and back but, they still can’t find a cure for the common hangover. In the simplest of terms, a hangover is a chemical consequence of imbibing in too many alcoholic drinks. Other than abstaining or simply cutting back, there really is no fool proof way to avoid the dreaded hangover. Some hangovers are mild and some are monsters. Then there is that one friend that we all have (that we all hate, it’s ok to admit it,) that is somehow genetically immune from hangovers.
Stay Informed: Sign up here for the Distillery Trail free email newsletter and be the first to get all the latest news, trends, job listings and events in your inbox.
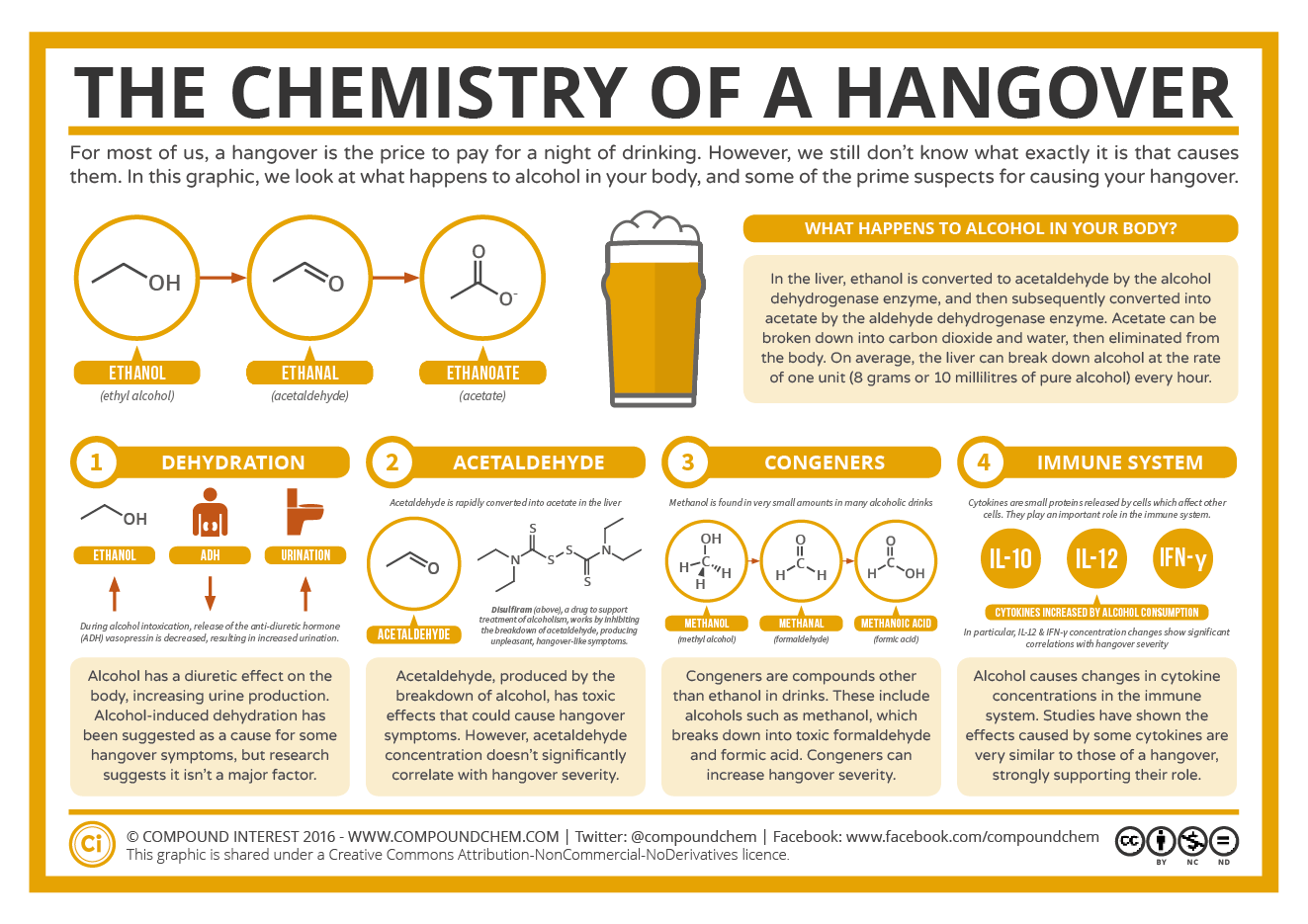
In this graphic, we look at what happens to alcohol in your body, and some of the prime suspects for causing your hangover.
-
Dehydration
Alcohol has a diuretic effect on the body, increasing urine production. Alcohol-induced dehydration has been suggested as a cause for some hangover systems, but research suggests it isn’t a major factor.
-
Acetaldehyde
Acetaldehyde, produced by the breakdown of alcohol, has toxic effects that could cause hangover systems. However, acetaldehyde concentration doesn’t significantly correlate with hangover severity.
-
Congeners
Congeners are compounds other than ethanol in drinks. These include alcohols such as methanol, which breaks down into toxic formaldehyde and formic acid. Congeners can increase hangover severity.
-
Immune System
Alcohol causes changes in cytokine concentrations in the immune system. Studies have shown the effects caused by some cytokines are very similar to those of a hangover, strongly supporting their role.
Some studies have suggested that the problem may lie with compounds other than alcohol in the drinks you were drinking the night before. Most alcoholic drinks will contain a whole range of other chemical compounds as well as ethanol, and these compounds are generally referred to as congeners. Different drinks have different levels of congeners; for example, brandy, red wine and whiskey have a much higher congener level than drinks such as beer, vodka or gin. It’s suggested that higher congener levels could increase the severity of hangover symptoms the next day.
Potential major players when it comes to congeners are alcohols other than ethanol. These are present in much lower quantities, but can include methanol. Whether you’re a chemist or not, you’ve likely heard of methanol, and you might be surprised to learn that it’s actually found in small quantities in most alcoholic drinks, as a byproduct of distillation or brewing processes. Though methanol is dangerous to ingest in large amounts, it’s not problematic in the amounts found in these alcoholic drinks – though it may make a contribution to hangover severity.
Head to Head Testing
Unlike many science experiments where you might have a test group and a control group it’s kind of difficult when it comes to drinking. Let’s say test group A is given the real juice and the control group B is given the fake juice. After a few hours, the subjects in the two groups are going to know which group got the real whiskey and which one got the iced tea. By the time morning roles around there will be no question which group was the placebo group.
We are not going to pretend to provide you with a definitive answer here. We are simply trying to share some of the common chemicals that are believed to make up the common hangover.
You can click on the Chemistry (101) of a Hangover Infographic to see it full size.
Please help to support Distillery Trail. Like us on Facebook and Follow us on Twitter.